|
Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm. The distance between the radii is 228 p m. The core electrons are said to shield the valence electrons from the full attractive forces of the protons in the nucleus. 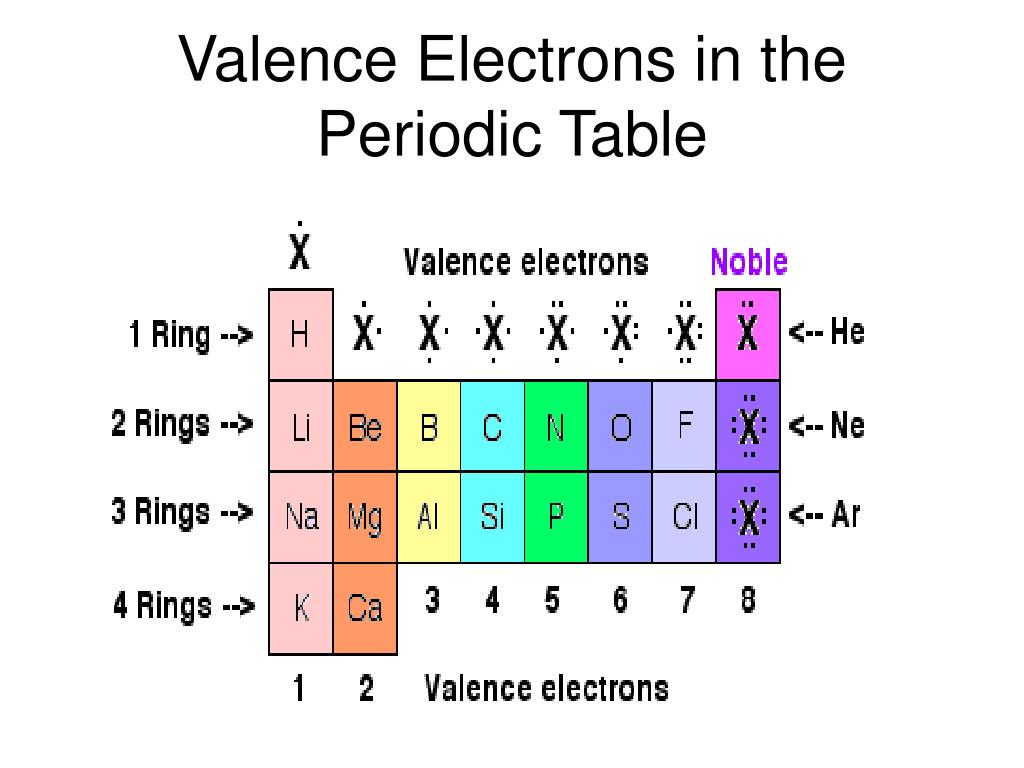 
Effective nuclear charge: the net positive charge from the nucleus that an electron can feel attractions from. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. The number of valence electrons of an element can be determined by the group (vertical column) number in the Periodic Table. Perfect your performance with periodicity Important Trend Terms. The distance between the radii is 198 p m. The second diatomic molecule is in a darker shade of green. Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. The Periodic Table of Elements categorizes like elements together. Therefore, the nucleus has less of a pull on the outer electrons and the atomic radii are larger. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. In a group, the valence electrons keep the same effective nuclear charge, but now the orbitals are farther from the nucleus. Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The general trend is that radii increase down a group and decrease across a period. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. 
\): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
